ColoAlert Stool Test
ColoAlert Stool Test
Advanced colon cancer screening test - trusted by patients across Europe
The ColoAlert stool test is an advanced screening test developed for the early detection of colon cancer. Unlike a conventional FIT or fecal immunochemical test, which can only detect hidden blood in stool, ColoAlert also analyzes samples for tumor DNA and additional biomarkers released by cancer cells. This allows ColoAlert to identify cancerous or precancerous growths at very early stages, when colon cancer is most treatable. The test uses molecular analysis to detect DNA changes in cells shed into the stool, providing patients with better early detection than blood-based screening alone.
| ✚ |
Advanced stool DNA testing – ColoAlert can detect colon cancer before symptoms appear, even at very early stages¹,² |
| ✚ |
Higher detection rate – compared with conventional screening tests (FIT)¹,² |
| ✚ |
Quick & convenient – sample collection completed in just 5 minutes with clear instructions in your test kit |
| ✚ |
Results in just a few days – your stool samples are processed in a certified laboratory and results delivred fast |
⟲ Free shipping and returns within Germany
Availability for pickups could not be loaded.

ColoAlert Is the Stool Test That Takes a Closer Look
FIT
ColoAlert
Mutated DNA
hDNA
An abnormal test result can indicate serious health problems and often requires a colonoscopy for further clarification and preventative healthcare.
About Half of Colon Cancer Cases Are Diagnosed at a Late Stage³
If the disease is detected early, the chances of a cure are very good. Unfortunately, half of all colon cancer cases are only found at an advanced stage.³ Reasons for this include the low uptake of colonoscopies⁴ and the limited accuracy of conventional stool tests.²
-

Colon Cancer Is Often Curable if Detected Early³
-

Conventional Stool Tests (FIT) Are Often Too Inaccurate²
-

Every Second Colon Cancer Diagnosis Is Made Too Late³
-

Reduced Chances of Recovery³ & More Difficult Therapy⁵
Let customers speak for us
from 167 reviewsDieser Test erspart mir schon seit Jahren eine Darmspiegelung!
Und letztes Jahr wurde - ebenfalls durch den Test - bei meinem Mann Darmkrebs im Frühstadium entdeckt. Nach einer OP und Chemotherapie in Tablettenform, welche er zu Hause durchführen konnte, gilt er heute schon wieder krebsfrei.
Ich empfehle diesen Test immer wieder Freunden und Bekannten!
Wer eine Alternative zur Coloskopie als Darmkrebs-Prophylaxe sucht, rate ich zum ColoAlert-Test. Neben dem iFOBT erfährt man, ob im Darm OncoGene nachgewiesen werden können, die bei einer Carcinom-Entwickung ein aggressives Wachstum voraus sagen. Der Test ist so einfach wie der iFOBT alleine und wird nur zu Hause vorgenommen. - Die Kommunikation mit dem European Oncology Lab - Team kann ich nur als hervorragend bewerten!
Es ging super schnell, innerhalb weniger Tage habe ich mein Ergebnis erhalten. Die Erklärung des Befundes war detailliert und verständlich. Ich habe ein besseres Gefühl. Vielen lieben Dank. Ich habe diesen Test weiter empfohlen und ihn auch für meine Mutter bestellt.
Mir gefällt vor allem die schnelle und leicht verständliche Auswertung. Fühle mich jetzt viel sicherer.
Das Testmaterial war super, aber der Rückversand war etwas umständlich, weil ich aus Spanien versenden musste. Insgesamt aber sehr zufrieden.
Das ist bereits mein zweiter Test und wieder war alles reibungslos. Klare Empfehlung meinerseits!
Rasche und sehr gute verständliche Erklärung der Untersuchungsergebnisse! Einfach gesamtbildlich betrachtet hervorragend und unbedingt weiterzuempfehlen! Die Weiterleitung an meine Krankenkasse ist am Laufen!
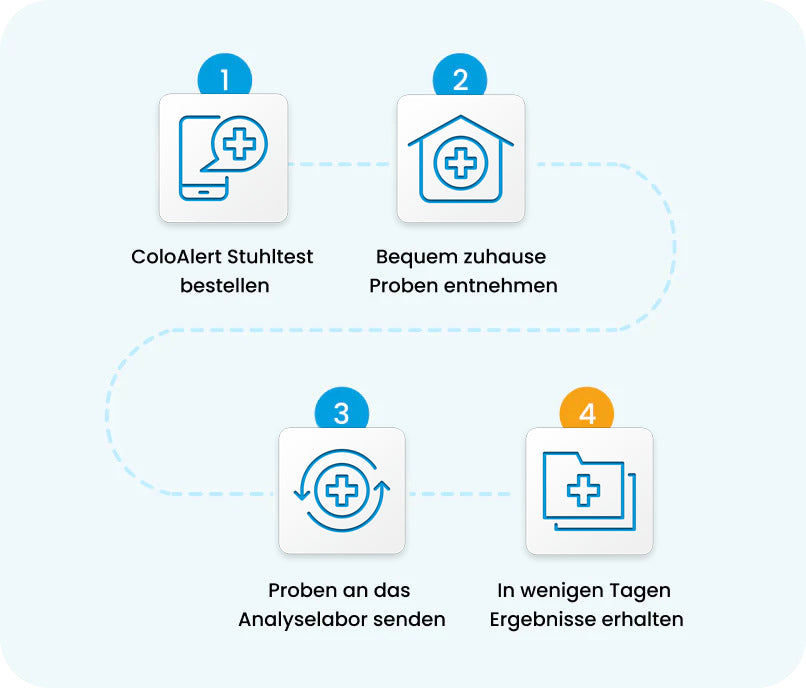
Colon Cancer Screening Is That Easy with the ColoAlert Stool Test
-

Order Your
ColoAlert Stool Test
-

Collect Samples
Comfortably at Home -

Return Samples to
Our Laboratory -

Receive Results Within
a Few Days
Frequently Asked Questions
For Whom Is the ColoAlert Colon Cancer Screening Test Suitable?
ColoAlert is generally suitable for patients aged 45 and over. Colonm cancer risk increases with age, and regular colorectal cancer screening from this age can help save lives. In certain cases – for example, if a family history of colon cancer or other risk factors are present – ColoAlert may also be advisable at a younger age after consultation with a physician. Patients with a family history of polyps or hereditary conditions should pay particular attention to screening. At every age, knowing your risk helps you make the right decision.
However, ColoAlert is not suitable for patients with irritable bowel syndrome, chronic inflammatory bowel conditions such as ulcerative colitis or Crohn's, as well as those with familial adenomatous polyposis (FAP) or hereditary non-polyposis colorectal cancer (HNPCC).
What Is ColoAlert? What Does the ColoAlert Stool Test Detect in the Laboratory?
ColoAlert is a next-generation colorectal cancer screening test. In the laboratory, ColoAlert analyzes your stool sample for four biomarkers: occult blood, two cancer gene marker (KRAS+BRAF) and one additional markers (hDNA). This approach allows ColoAlert to detect colorectal cancer at very early stages, when the condition may not yet cause bleeding or other symptoms.
Mainz Biomed, the diagnostics company behind ColoAlert, develops and optimizes screening tests for the detection of colon cancer. These additional markers increase the sensitivity of ColoAlert compared to a standard FIT – meaning colon cancer is more likely to be detected early. Each of the stool samples received is processed in our laboratory with strict quality standards. How does ColoAlert work in practice? The sample you send to the lab is analyzed using molecular diagnostic techniques that can detect DNA shed by abnormal cells, making the test far more thorough than blood-only methods.
Has the ColoAlert Screening Test Been Evaluated in Clinical Studies?
All screening tests are subject to a certain error rate. In diagnostics, accuracy is represented by sensitivity (proportion of true positive results) and specificity (proportion of true negative results).
The sensitivity of ColoAlert is 85%, and the specificity is 92%, according to several independent clinical studies. This means ColoAlert can reliably detect colorectal cancer while minimizing false positive results. In one study, ColoAlert achieved better results than the occult blood and M2-PK tests that were also evaluated.¹,² ColoAlert colon cancer screening is processed in our laboratory and all samples are analyzed under strict quality standards. Patients consistently leave positive reviews about the reliability and speed of their results.
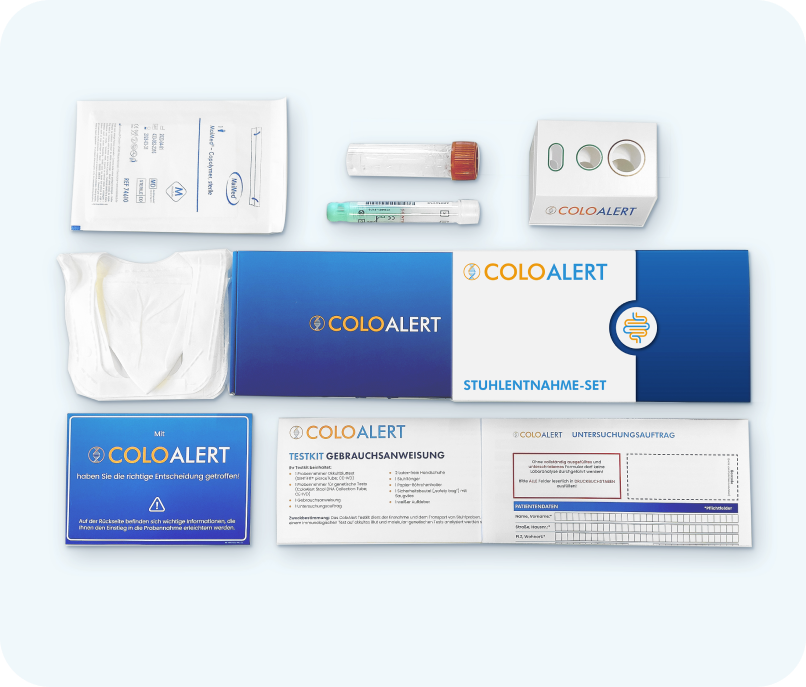
How Do I Collect the Sample with the ColoAlert Test Kit?
Collecting a stool sample for the test is quick and simple – taking only about 5 minutes. You will find detailed, clear instructions in your ColoAlert test kit that guide you step by step through the sample collection process. The kit includes everything you need: collection materials, return packaging, and clear instructions. You can also use the video tutorial for assistance. Once your stool samples are collected, simply return them to the lab using the prepaid envelope in your kit.
References
1) Krammes, L., Mahmood, HA, Frondorf, FMB, Scholz, CF, Becker, P., Maharjan, S., Sever, AE, Garapati, SV, Balasubramaniam, A., Knabe, MJ, Eidens, MR, Dollinger, MM (2025). State-of-the-Art Colorectal Cancer and Advanced Precancerous Lesion Screening: a Multitarget Stool DNA Test, Clinical Laboratory, 71(1), 34-39. doi.org/10.7754/Clin.Lab.2024.240620 ; Dollinger, MM, Behl, S., & Fleig, WE (2018). Early Detection of Colorectal Cancer: a Multi-Center Pre-Clinical Case Cohort Study for Validation of a Combined DNA Stool Test. Clinical Laboratory, 64(10), 1719-1730. doi.org/10.7754/Clin.Lab.2018.18052 2) Gies, A., Cuk, K., Schrotz-King, P., & Brenner, H. (2018). Direct Comparison of Diagnostic Performance of 9 Quantitative Fecal Immunochemical Tests for Colorectal Cancer Screening. Gastroenterology, 154(1), 93–104. doi.org/10.1053/j.gastro.2017.09.018 3) Robert Koch Institute and the Association of Population-Based Cancer Registries in Germany (2023). Cancer in Germany for 2019/2020, 14th edition. 4) Steffen, A., Holstiege, J., Hagen, B., Akmatov, MK, Bätzing, J. (2020). Utilization of colorectal cancer screening from 2009 to 2018: an inventory based on nationwide billing data from contracted physicians. Central Institute for Statutory Health Insurance Physicians in Germany (Zi). Healthcare Atlas Report No. 20/02. DOI: 10.20364/VA-20.02. 5) Guidelines Program Oncology (German Cancer Society, German Cancer Aid, AWMF) (2019). S3 Guideline Colorectal Carcinoma, Long Version 2.1, AWMF Registration Number: 021/007OL [ https://www.leitlinienprogramm-onkologie.de/leitlinien/kolorektales-karzinom/ Accessed: March 15, 2024]




