
The modern stool test for the early detection of colorectal cancer
ColoAlert not only detects blood in stool like conventional stool tests, but also tumor-related DNA changes. This makes colorectal cancer visible in its early stages – discreetly, safely, and conveniently from home.
Early detection through tumor DNA detection
Multiple clinically proven detection rates
Convenient and precise preventative care from home
Reimbursable for privately insured individuals
ColoAlert in the media





Every second diagnosis of colorectal cancer is made in late stages.
Every eight minutes, someone in Germany is diagnosed with colorectal cancer. If the disease is detected early, the chances of a cure are very good . Unfortunately, half of those affected only receive the diagnosis in an advanced stage. Reasons for this include the low uptake of colonoscopies and the limited detection rate of conventional stool tests.
-

If detected early, colon cancer is often curable.
-

Conventional stool tests are often too inaccurate.
-

Every second diagnosis is made in the late stage.
-

Reduced chances of recovery & more difficult therapy
Der Stuhltest, der ganz genau hinschaut
The ColoAlert stool test was developed to prevent late diagnoses of colorectal cancer. Unlike conventional stool tests (iFOBT), which only check for hidden blood in the stool, ColoAlert also analyzes samples for tumor DNA . This allows colorectal cancer to be detected significantly earlier. Tumor DNA consists of genetic markers released into the stool by tumors in the colon – and can be detected at a much earlier stage of the disease than blood.
⟲ Free shipping and returns within Germany
✓ Reimbursable for privately insured individuals
€257.98
Split

Let customers speak for us
from 136 reviewsDieser Test erspart mir schon seit Jahren eine Darmspiegelung!
Und letztes Jahr wurde - ebenfalls durch den Test - bei meinem Mann Darmkrebs im Frühstadium entdeckt. Nach einer OP und Chemotherapie in Tablettenform, welche er zu Hause durchführen konnte, gilt er heute schon wieder krebsfrei.
Ich empfehle diesen Test immer wieder Freunden und Bekannten!
Wer eine Alternative zur Coloskopie als Darmkrebs-Prophylaxe sucht, rate ich zum ColoAlert-Test. Neben dem iFOBT erfährt man, ob im Darm OncoGene nachgewiesen werden können, die bei einer Carcinom-Entwickung ein aggressives Wachstum voraus sagen. Der Test ist so einfach wie der iFOBT alleine und wird nur zu Hause vorgenommen. - Die Kommunikation mit dem European Oncology Lab - Team kann ich nur als hervorragend bewerten!
Es ging super schnell, innerhalb weniger Tage habe ich mein Ergebnis erhalten. Die Erklärung des Befundes war detailliert und verständlich. Ich habe ein besseres Gefühl. Vielen lieben Dank. Ich habe diesen Test weiter empfohlen und ihn auch für meine Mutter bestellt.
Mir gefällt vor allem die schnelle und leicht verständliche Auswertung. Fühle mich jetzt viel sicherer.
Das Testmaterial war super, aber der Rückversand war etwas umständlich, weil ich aus Spanien versenden musste. Insgesamt aber sehr zufrieden.
Das ist bereits mein zweiter Test und wieder war alles reibungslos. Klare Empfehlung meinerseits!
Rasche und sehr gute verständliche Erklärung der Untersuchungsergebnisse! Einfach gesamtbildlich betrachtet hervorragend und unbedingt weiterzuempfehlen! Die Weiterleitung an meine Krankenkasse ist am Laufen!
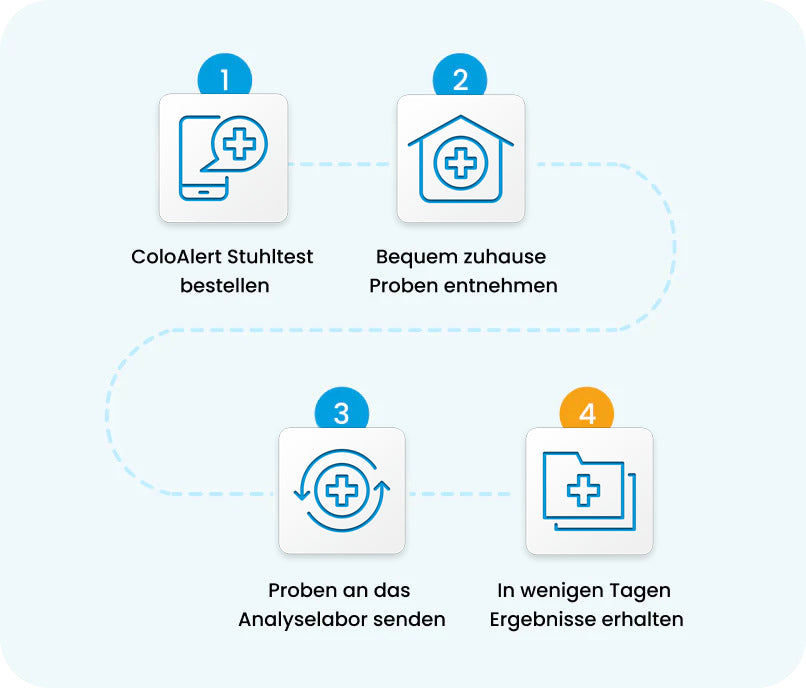
Colon cancer screening is that easy with the ColoAlert stool test.
-

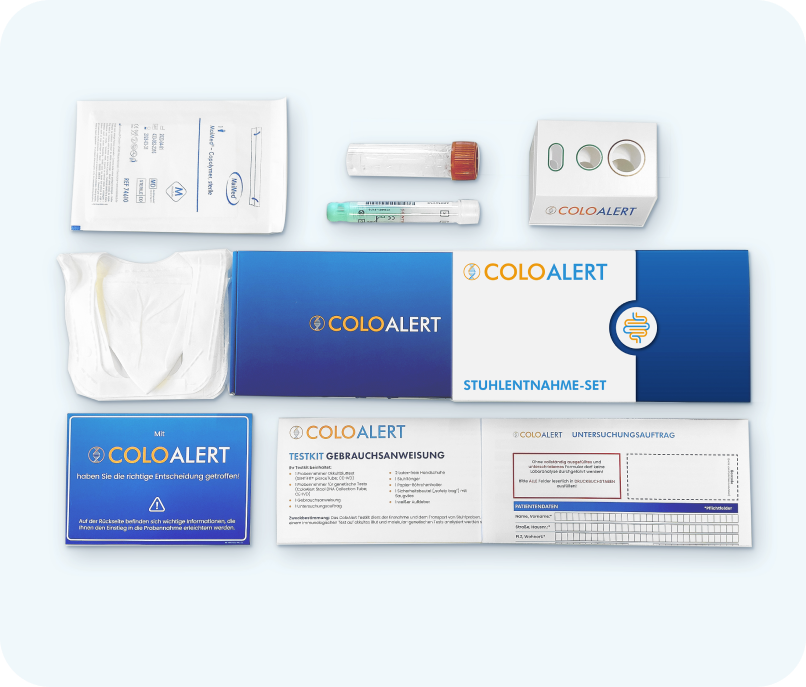
ColoAlert stool test
order -

Comfortable at home
Take samples -

Samples to us
return -

In a few days
Results received
Frequently Asked Questions
For whom is the ColoAlert stool test suitable as a preventative measure?
ColoAlert is generally suitable for people aged 45 and over. Public acceptance of preventive measures like the ColoAlert test is increasing, especially compared to occult blood tests (iFOBTs). In certain cases—for example, if additional risk factors are present—use may also be advisable outside this age range after consultation with a doctor.
However, ColoAlert is not suitable for people with irritable bowel syndrome, chronic inflammatory bowel diseases such as ulcerative colitis or Crohn's disease, as well as for those with familial adenomatous polyposis (FAP) or hereditary non-polyposis colorectal cancer (HNPCC).
What is the sample tested for in the laboratory during the ColoAlert stool test?
In addition to detecting occult blood, ColoAlert analyzes three further tumor markers. The molecular genetic analysis examines the stool sample for the presence of suspiciously high amounts of human DNA and genetic mutations. The diagnostics company Mainz Biomed develops and optimizes tests for the early detection of colorectal cancer to increase the efficiency and user-friendliness of screening tests. These additional features increase the detection rate for colorectal cancer, the so-called sensitivity (proportion of positive test results among those affected). Mainz Biomed specializes in cancer screening and offers innovative solutions for the early identification of cancers.
Has the ColoAlert stool test been subjected to clinical trials?
All measurement methods are subject to a certain error rate. In diagnostics, the error rate is represented by sensitivity (proportion of true positive results) and specificity (proportion of true negative results).
The sensitivity of the ColoAlert colorectal cancer test (Combined DNA stool assay) is 85% and the specificity is 92%, according to several independent clinical studies. Mainz Biomed, the company behind ColoAlert, developed the test with advanced features to optimize user-friendliness and screening efficiency. In one study, ColoAlert achieved better results in terms of sensitivity and specificity than the occult blood and M2-PK tests that were also evaluated.




